If you have any question, please feel free to email us. We will touch with you as soon as possible.
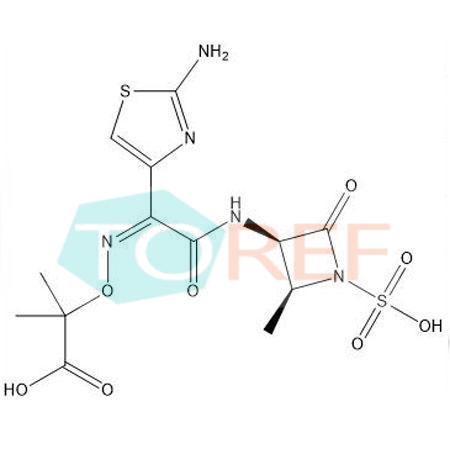
product name:Aztreonam Impurity 59
CAS:N/A
Product Code:REF-A04048
MF:C13H17N5O8S2
MW:435.43
Purity:98.64%
 product name:Aztreonam Impurity 59
product name:Aztreonam Impurity 59
CAS:N/A
Product Code:REF-A04048
MF:C13H17N5O8S2
MW:435.43
Purity:98.64%
Product nature:Customer customization
Unit:mg
Min. Order:5mg
Supply Ability:1000
Location:china
color/storage temp:White SolidStore at -20℃
Related Names:Aztreonam Impurity 59,Aztreonam Impurity 59Standard,Aztreonam Impurity 59Reference
TOSUN PHARM was established in 1999. In China, it is a collection of drugs, APIs, reference listed drugs, impurities, excipients, intermediates import and export, import registration services, generic drug research and development services, innovative drugs and high-end FDF technology transfer, A group company integrating marketing, academic promotion and cooperative production. With a global procurement, R&D and marketing network, it can quickly meet the needs of product R&D, production and market sales of Chinese and global pharmaceutical companies.

Synthesis of Aztreonam Impurity 59
Complex Impurity Of Aztreonam Impurity 59
Chemical Standards of Aztreonam Impurity 59,
Characterization Of Unknown Impurities of Aztreonam Impurity 59
Structure Profiling of Aztreonam Impurity 59
Identification of Aztreonam Impurity 59
Isolation & Purification of Impurity of Aztreonam Impurity 59
Reference Standards Aztreonam Impurity 59
Research Chemical Aztreonam Impurity 59
Drug substance Aztreonam Impurity 59
Impurity Standards Aztreonam Impurity 59
Reference Standards Aztreonam Impurity 59
Work Standards Aztreonam Impurity 59
Complex Impurity Of Chemical Standards of Aztreonam Impurity 59
Hot Tags:Aztreonam Impurity 59, China, suppliers, manufacturers, factory, customized, price, price list, in stock.
Tel:+86-020-61855200-673
Fax:+86-020-66392525
Email:info@upharm.cn
Address:12th floor, No. 181, Kexue Avenue, Huangpu District, Guangzhou, China